When Vimal Selvaraj’s uncle first imported Holstein semen to start his dairy farm in India in the early 1990s, he was hailed as a revolutionary on the cutting edge of agriculture. His first generation of cattle crosses between Holsteins and Sahiwals, a native Indian breed, seemed to carry the best traits of both: the Holsteins’ high milk production, and the Sahiwals’ disease resistance and extreme weather tolerance.
But after a few generations, as the cattle carried more and more Holstein genetics, cows became sicker and sicker. They suffered from mastitis, parasitic infestations and other diseases that the Sahiwal had tolerated. Just 10 years after that first Holstein cross, the farm went bankrupt.
“This extreme focus on milk-production traits has had beneficial impacts, but it has had collateral damage as well,” said Selvaraj, associate professor of animal science at Cornell University. “Animal production is driven by industry, and industry is driven by profit. Many people are thinking two to three years ahead, not 20 years ahead.”
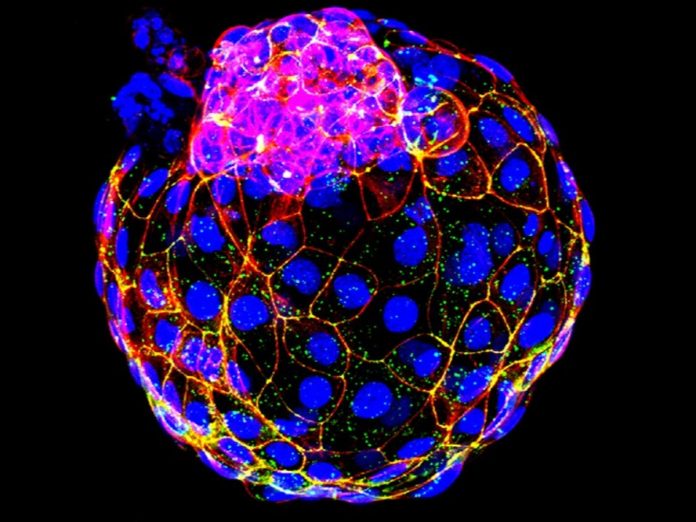
Today, Selvaraj is working to preserve the genetic diversity of fast-disappearing native cattle breeds via gene banking, and his lab has made a breakthrough that will enable long-term storage and reproduction of cattle stem cells – cells that could be used in the future to clone breeds that have gone extinct, or that have been so heavily crossed with other breeds that they’ve lost their strongest traits.
Their findings were reported in “Efficient Induction and Sustenance of Pluripotent Stem Cells from Bovine Somatic Cells,” published in Biology Open on Nov. 1. First authors are Viju Vijayan Pillai, a former doctoral student under Selvaraj and now a veterinary resident at Purdue University, and Prasanthi Koganti, a postdoctoral associate in Selvaraj’s lab.
Traditionally, farms kept a few high-performing bulls to impregnate their cows, swapping bulls with neighbors to prevent inbreeding. In the 1980s, though, most U.S. farmers moved to artificial insemination, drawn by not having to keep large, dangerous bulls, and by being able to choose the most genetically superior males from across the country. But everyone was looking for the same traits, and the result is that now, 99% of U.S. Holstein cattle are descended from just two bulls, both born in the 1960s.
The problem is spreading across livestock worldwide: according to the United Nations, almost 100 livestock breeds went extinct between 2000 and 2014, and another 17% of global livestock breeds are at risk of extinction. That could have severe consequences as climate change increases temperatures, allows disease and insects to spread to new territories and makes traits like hardiness and disease resistance ever-more important.
“If a new disease or organism suddenly comes in and if the animals are extremely susceptible – and they are more or less homogeneous – then we have a very, very serious problem on our hands,” Selvaraj said.
Similar to the Norwegian-led global seed bank, Selvaraj advocates for a global livestock stem-cell repository. The United Nations and the American Veterinary Medical Association are among those calling for a livestock genome bank. But preserving livestock genetics is much more complicated than seeds. Globally, 64 countries have established genome banks of some kind, including the U.S.; however, these banks are primarily composed of cryopreserved bull semen. This strategy is good, but stem cell preservation is much better, Selvaraj said. Stored semen is a finite resource, and it only provides half the genome: Semen would require cross-breeding with a living cow, while stem cells could be used to clone a preserved, pure-bred native breed.
Previous efforts to generate cattle stem cells have had mixed results, because the science was fully based on mouse and human research, Selvaraj said. His team sequenced gene expressions in approximately 400 cattle blastocyst embryos. Using a systems biology approach, they identified the top pathways important for the process of stem cell self-renewal, and determined the conditions needed to prevent the stem cells from differentiating into other cell types.
“Using stem cells, you can preserve the full genome and then regenerate the animal at any future point in time,” Selvaraj said. “Stem cells undergo self-renewal and are immortal, so you can use some and refreeze them. This provides an inexhaustible store that you can use into the future.”